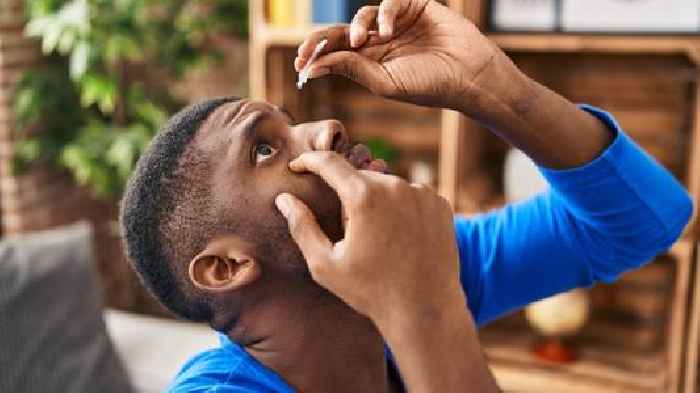
ViewThe Food and Drug Administration announced this week two additional eye drop brands have initiated voluntary recalls as the products lack sterility.
Pharmedica USA recalled two lots of Purely Soothing 15% MSM Drops. The eye drops are used as an anti-inflammatory and assist with eye swelling and irritation.
The eye...
More eye drops recalled for lacking sterility
Newsy
0 shares
1 views
You might like
Related news coverage
68 injuries, 4 eyeball removals tied to recalled EzriCare eye drops
Newsy
ViewThe Centers for Disease Control and Prevention is now investigating 68 cases of eye injuries tied to recalled eye..
Advertisement
More coverage
Children's toys recalled after 2 deaths
Newsy
ViewMore than 3 million Calico Critters flocked animal figures and set have been recalled.
According to th Consumer..